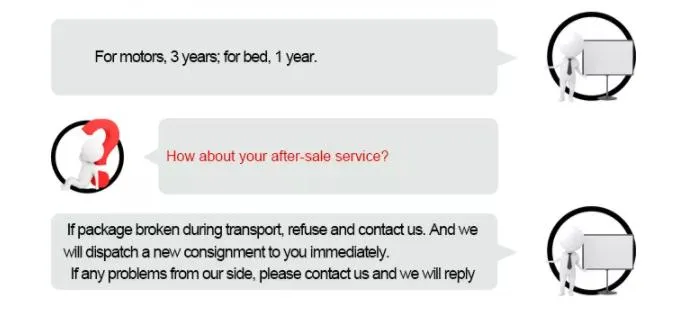
1 / 5











| Product Name | Code | Specification | Note |
|---|---|---|---|
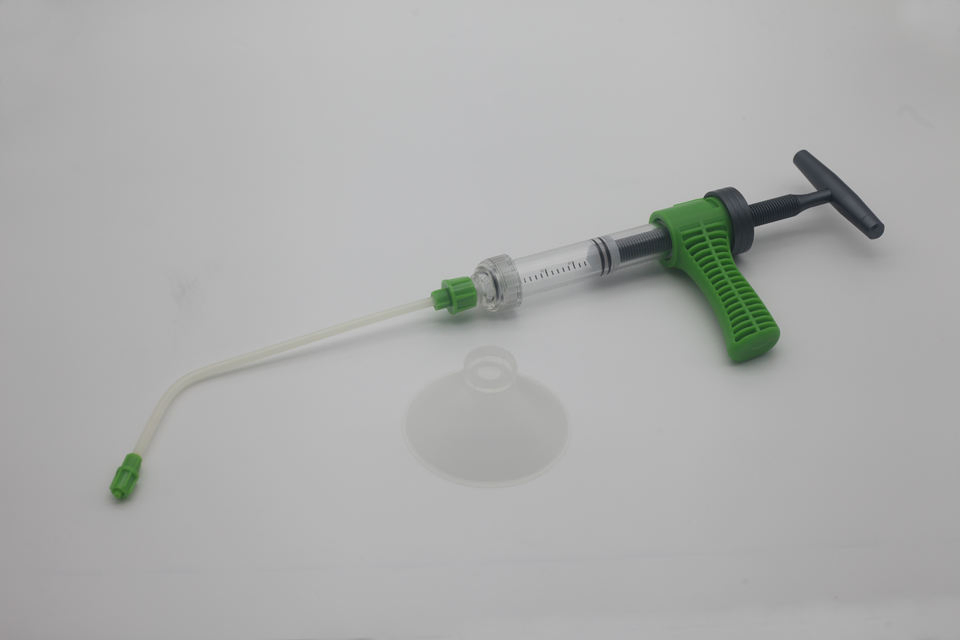
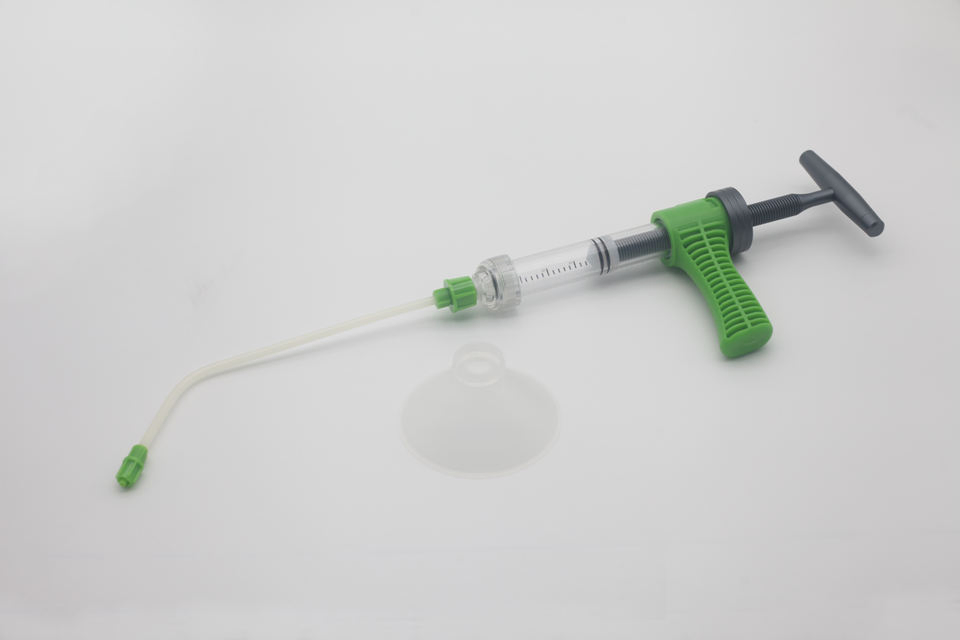
| Syringe injector(for Lumbar) | 030700004 | GTZ01 | / |
| Licence Number | CE, ISO | ||
*All the prices are only for reference. Exact price will be based on the latest quotation.