



1 / 5











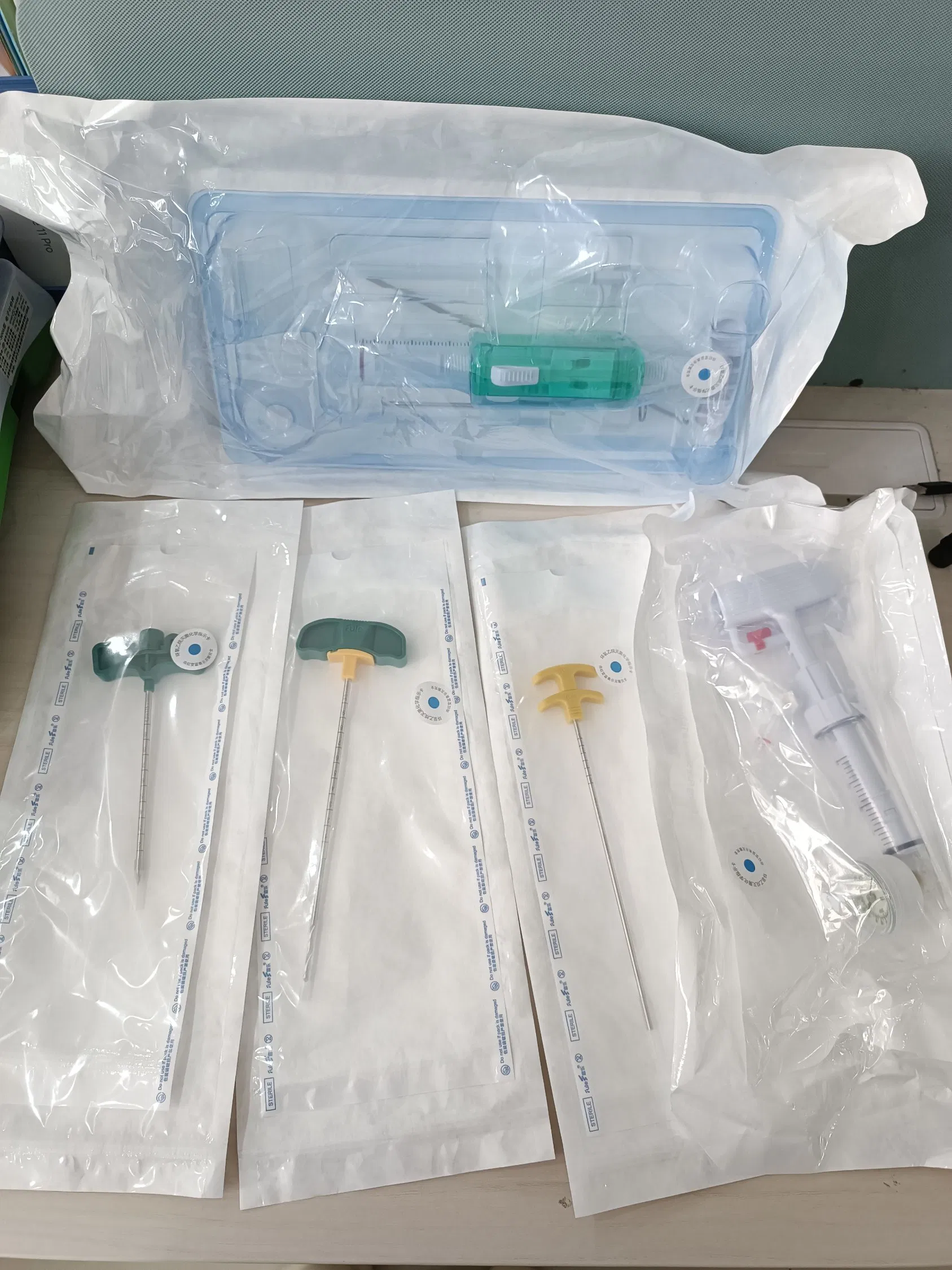
| Product Name | Code | Specification | Note |
|---|---|---|---|
| Bone Cement Applier (for Thoracic Vertebra) | 030436030 | ZT-III Φ3.0×200 | Plain Top |
| 030436130 | ZT-III Φ3.0×200 | Directional | |
| 030437030 | ZT-III Φ3.0×200 | Dentiform | |
| Kyphoplasty Balloon | 030200010 | QN-I 10 | Lumbar Vertebra |
| 030200015 | QN-I 15 | Lumbar Vertebra | |
| 030200020 | QN-I 20 | Lumbar Vertebra | |
| 030200110 | QN-I 10 | Thoracic Vertebra | |
| 030210215 | QN-II 15 | Lumbar Vertebra | |
| 030210220 | QN-II 20 | Lumbar Vertebra | |
| 030210210 | QN-II 10 | Thoracic Vertebra | |
| Balloon Inflation Pump | 030300030 | YLB-30 | / |
| 030300025 | YLB-25 | / | |
| 030300020 | YLB-20 | / | |
| Percutaneous Access Device (for Lumbar) | 030422042 | ZT-I Φ4.2×140 | / |
| 030422030 | ZT-I Φ3.0×120 | / | |
| Guide Wire (for Lumbar) | 030421155 | ZT-II Φ1.5×270 | / |
| Expansion Cannula (for Lumbar) | 030428042 | ZT-II Φ4.2×130 | Common |
| 030428242 | ZT-II Φ4.2×150 | Directional |
*All the prices are only for reference. Exact price will be based on the latest quotation.



